FDA Panel Says Pfizer COVID Booster OK For Older People And Those At High Risk : Coronavirus Updates : NPR

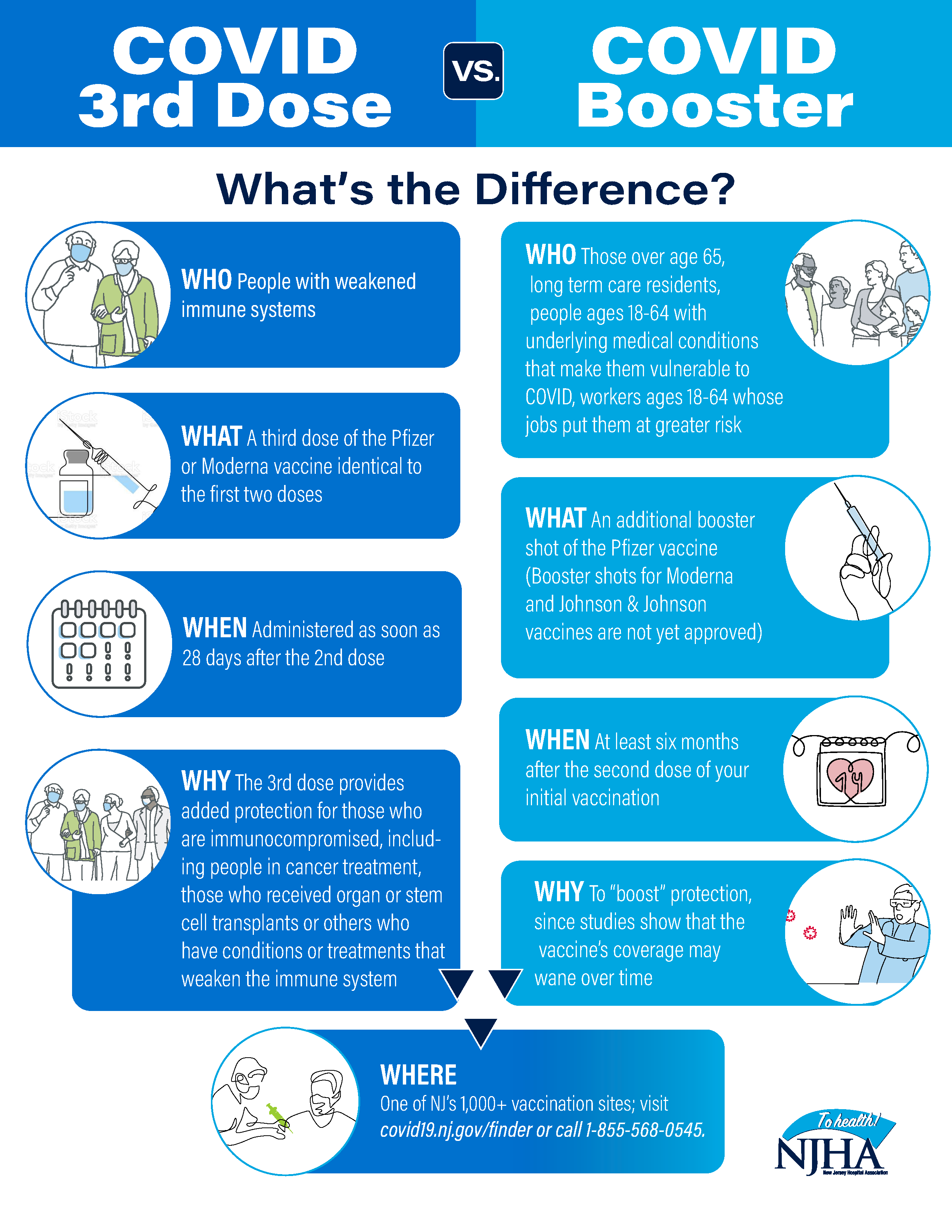
COVID-19 vaccines – The difference between the third dose and a booster dose – Easy Read | Australian Government Department of Health and Aged Care
Pfizer, BioNTech say bivalent COVID-19 booster shot performs better against BA.5 omicron subvariant - ABC News

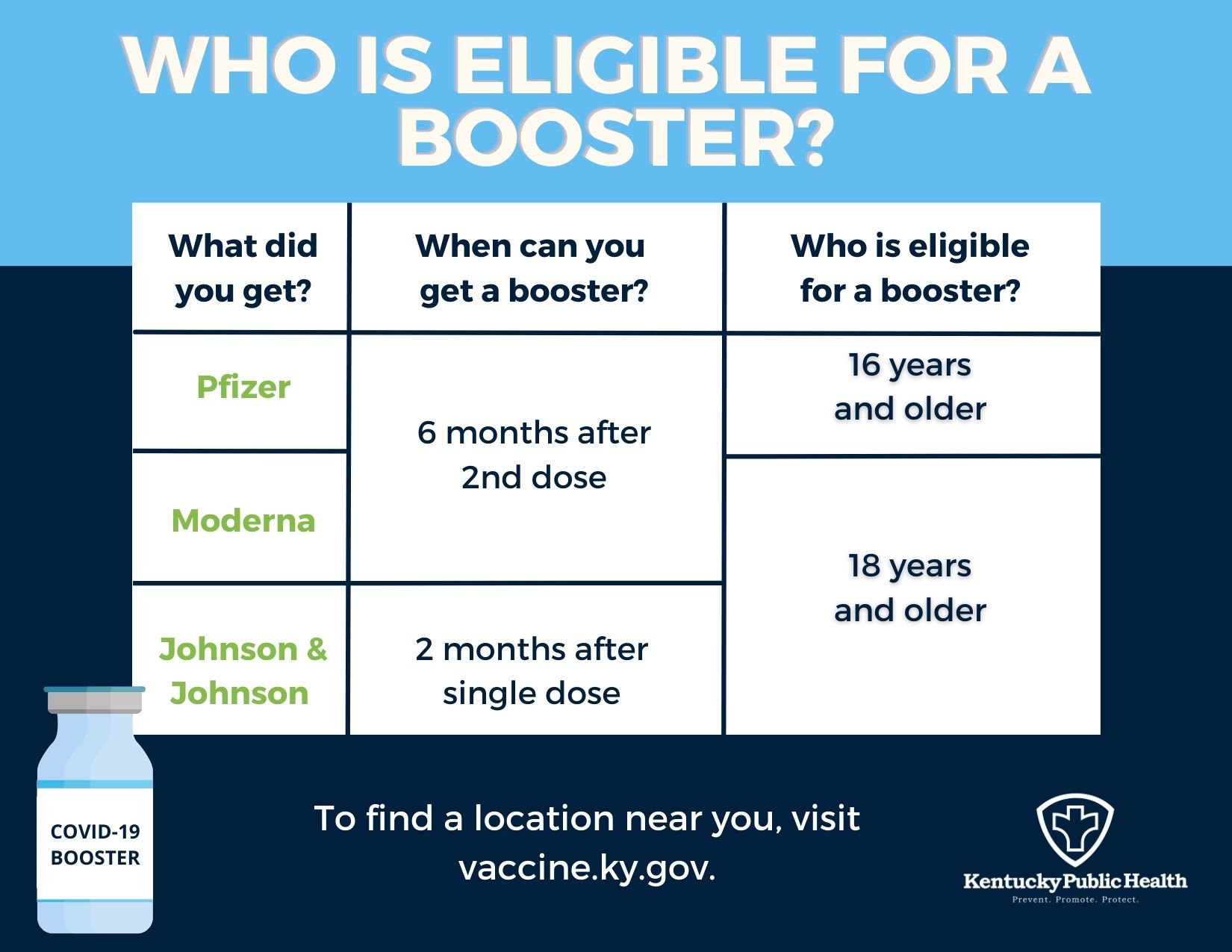
CDC on X: "CDC recommends that certain populations receive a booster shot of Pfizer-BioNTech's #COVID19 Vaccine at least 6 months after completion of their 2-dose Pfizer-BioNTech vaccine series. See full details: https://t.co/77CTFv1g4m.

Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA
Booster Shots and Additional Doses for COVID-19 Vaccines — What You Need to Know | Johns Hopkins Medicine









.png)